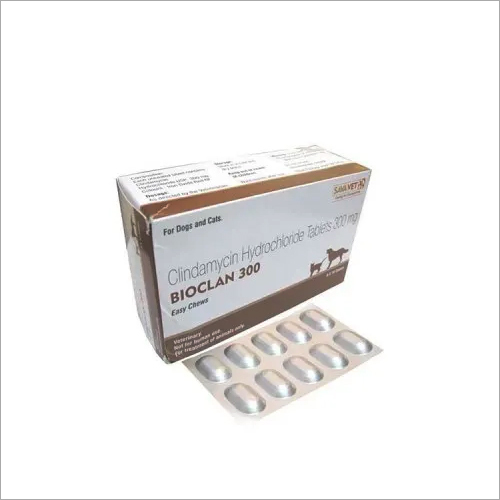
BIOCLAN 300MG TABLETS-CLINDAMYCIN
Product Details:
- Medicine Type Veterinary Raw Materials
- Physical Form Tablets
- Ingredients Chemicals
- Recommended For Dogs Cats
- Storage Instructions Cool & Dry Place
- Click to View more
BIOCLAN 300MG TABLETS-CLINDAMYCIN Price And Quantity
- 154 INR/Box
- 1 Box
BIOCLAN 300MG TABLETS-CLINDAMYCIN Product Specifications
- Tablets
- Cool & Dry Place
- Chemicals
- Dogs Cats
- Veterinary Raw Materials
BIOCLAN 300MG TABLETS-CLINDAMYCIN Trade Information
- 100 Box Per Day
- 9 Days
- 10 tab
- Western Europe Australia Middle East Central America South America Asia Eastern Europe North America Africa
- All India
Product Description
DESCRIPTION:
Clindamycin inhibits bacterial protein synthesis at the ribosomal (50s sub-unit) level.
Absorption:
Clindamycin hydro chloride is rapidly absorbed from the canine gastrointestinal tract following oral administration. Effective clindamycin antibacterial serumlevels are reached within 30 minutes following administration of the therapeutic dose.
Serum values:
Therapeutic serum levels of clindamycin can be maintained by oral administration of 5.5 mg/kg body weight every 12 hours or 11mg/kg bodyweight every 24 hours; peak serum concentrations are on average reached 75 minutes after oral administration. The biological plasma half-life of clindamycin in the dog is approximately 5 hours. No accumulation of bio activity has been observed in dogs after several oral administrations.
Metabolism and Excretion:
Extensive research of the metabolism and excretion pattern of clindamycin shows that the parent molecule as well as bioactive and bio-inactive metabolites is excreted via the urine and faeces. Nearly all bio activity in the serum following oral administration is due to the parent molecule (clindamycin).
INDICATIONS:
Clindamycin uncoated tablet are indicated for use in dogs as follows:
For the treatment of infected wounds and abscesses,and infected mouth cavity and dental infections, caused by or associated with Staphylococcus spp, Streptococcus spp (except Streptococcus faecalis),Bacteroides spp, Fuso bacterium necrophorum, and Clostridium perfringens. To help provide antimicrobial cover during dental procedures.
For the treatment of superficial pyoderma associated with Staphylococcus intermedius.
For the treatment of osteomyelitis, caused by Staphylococcus aureus.
Before Clindamycin therapy is initiated, the involved pathogens should be identified and sensitivity to clindamycinestablished.
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese



 Call Me Free
Call Me Free