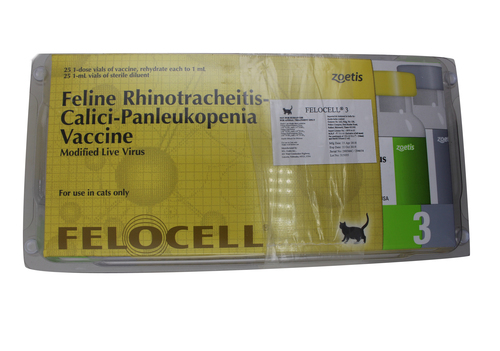

FELOCELL -3-felin
Product Details:
- Medicine Type Veterinary Raw Materials
- Physical Form Liquid
- Ingredients Chemicals
- Recommended For Pig Goat Dogs Cats Pets
- Storage Instructions 2-8
- Click to View more
FELOCELL -3-felin Price And Quantity
- 900 INR/Box
- 1 Box
FELOCELL -3-felin Product Specifications
- Veterinary Raw Materials
- Chemicals
- 2-8
- Liquid
- Pig Goat Dogs Cats Pets
FELOCELL -3-felin Trade Information
- Cash in Advance (CID) Cash Advance (CA)
- 100 Box Per Day
- 9 Days
- 1 dose
- All India
Product Description
DESCRIPTION:
Fello cell 3 is for vaccination of healthy cats as an aid in preventing feline viralrhinotracheitis (FVR) caused by feline herpesvirus-1, feline respiratory disease caused by feline calicivirus (FCV), and feline panleukopenia (FPL)caused by feline parvo virus (FPV). Fello cell 3 contains attenuated strains of feline rhinotracheitis virus, calici virus, and panleukopenia virus (Johnson Snow Leopard strain), propagated on established cell lines. Fello cell 3 is packaged in freeze-dried form with inert gas in place of vacuum.
Directions
- General Directions:Vaccination of healthy cats is recommended. Aseptically re-hydrate the freeze-dried vaccine with the sterile diluent provided, shake well, and administer 1 mL intra muscularly or subcutaneously.
- Primary Vaccination:Healthy cats 12 weeks of age or older should receive 2 doses administered 3-4weeks apart. Cats vaccinated at less than 12 weeks of age should be re-vaccinated at 12 weeks of age.
- Re-vaccination: Annual re-vaccination with a single dose is recommended as recommended by the American Veterinary Medical Association and its Council on Biologic and Therapeutic Agents, the attending veterinarian should determine the frequency of re-vaccination based on the animal's lifestyle and risk of exposure.4
Precautions
- Store at2°-7°C. Prolonged exposure to higher temperatures and/or direct sunlight may adversely affect potency. Do not freeze.
- Use entire contents when first opened.
- Sterilized syringes and needles should be used to administer this vaccine. Do not-sterilize with chemicals because traces of disinfectant may inactivate the vaccine.
- Burn containers and all unused contents.
- Contains gentamicin as preservative.
- Vaccination of pregnant queens should be avoided.
- As with many vaccines, anaphylaxis may occur after use. Initial antidote of epinephrine is recommended and should be followed with appropriate supportive therapy.
This product has been shown to be efficacious in healthy animals. A protective immune response may not be elicited if animals are incubating an infectious disease,are malnourished or parasitized, are stressed due to shipment or environmental conditions, are otherwise immuno compromised, or the vaccine is not administeredin accordance with label directions.
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese



 Call Me Free
Call Me Free